In a real-life, textbook example of host-microbe interaction, 2 parallel arcs have dominated “recent” SARS-CoV-2 news—vaccines and variants. As we continue the race to expand our pharmaceutical arsenal, the virus continues to accrue variations in its genome that have caused a considerable amount of public concern and prompted questions about the impact of this evolving public health threat.
How do we accommodate for changes in the genome of a virus that, let’s be honest, we’re barely getting acquainted with in the first place? And what consequences does all this have on vaccine development, antibody response and global efforts to identify and prevent the spread of disease? Here we take a look at some noteworthy amino acid changes to the S protein of SARS-CoV-2 variants and dissect emerging research about the resultant antibody responses of vaccinated individuals.
How Frequently Is SARS-CoV-2 Mutating?
A typical SARS-CoV-2 virus accumulates 1-2 single-nucleotide mutations in its genome per month, which is ½ the rate of influenza and ¼ of the rate of HIV. Part of the reason that SARS-CoV-2 appears to be mutating more slowly is that, unlike most RNA viruses, coronaviruses have a novel exoribonuclease (ExoN) encoded in their genomes, which researchers suspect is correcting many of the errors that occur during replication. Genetic inactivation of this exonuclease in SARS-CoV and Murine coronavirus (MHV) increased mutation rates by 15-to-20-fold. And the molecular basis of this CoV proofreading complex is being investigated as a possible therapeutic target for SARS-CoV-2. Importantly, nucleotide deletions, unlike substitutions, cannot be corrected by this proofreading mechanism, which is a factor that may accelerate adaptive evolution to some degree.
S Protein Mutations in SARS-CoV-2 Variants
Depending on the specific mutation, and where in the genome the nucleotide substitution, addition or deletion occurs, mutations may be neutral, beneficial or harmful to an organism. SARS-CoV-2's spike (S) protein is 1273 amino acids long and is the main target of current COVID-19 vaccines, as well as those in development. It is the portion of the virus that recognizes and binds to host cellular receptors and mediates viral entry. SARS-CoV-2 is unable to infect host cells without it. Because of this, mutations in the S gene, particularly those that affect portions of the protein that are critical for pathogenesis and normal function (such as the receptor binding domain (RBD) or furin cleavage site) or those that cause conformational changes to the S protein, are of the greatest interest. If these changes are not recognized by “first-wave” antibodies, these mutations may provide an avenue for the virus to escape from immunity to the original SARS-CoV-2 strain.
Below is a brief introduction of the first reported SARS-CoV-2 mutation, D614G, which has now become common to nearly all sequenced SARS-CoV-2 genomes worldwide, followed by analysis of additional key S protein mutations of a number of identified SARS-CoV-2 variants: B.1.1.7, commonly dubbed the U.K. variant; B.1.351, also known as 501Y.V2 or the South African variant, P.1., also known as 501Y.V3 or the Brazilian variant; B.1.427 and B.1.429, also recognized as CAL.20C or the California variant; B.1.526, or the New York variant and multiple lineages of variants that contain mutations at amino acid position 677 .
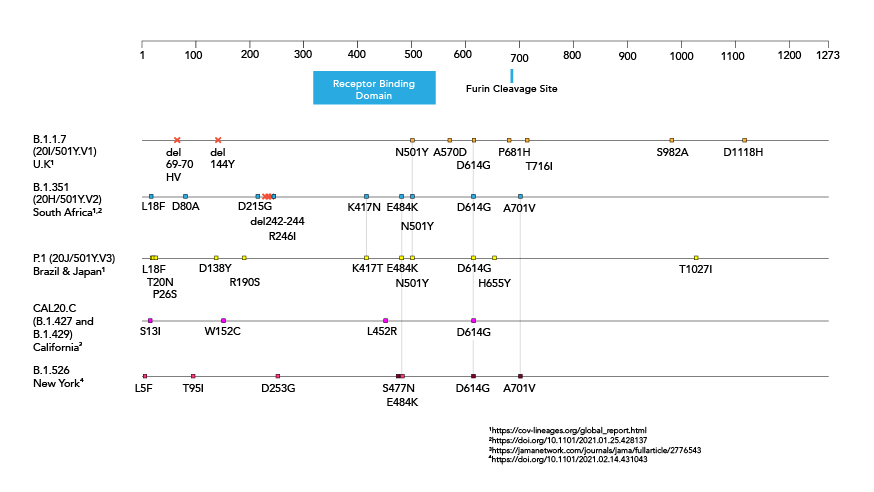
[Refer to the picture above]
Amino Acid Changes in the Spike (S) Protein in SARS-CoV-2 Variants
Amino acid changes in the spike protein of SARS-CoV-2 variants.
Amino acid changes in the spike protein of B.1.1.7, B.1.351, P.1, B.1.427/B.1.429 and B.1.526 SARS-CoV-2 variants.
Source: American Society for Microbiology
D614G
Initial reports that a mutation had been identified in the SARS-CoV-2 genome began circulating in March 2020, and by the end of June, D614G, which constitutes replacement of aspartate (D) with glycine (G) at the 614th amino acid of S protein, was found in nearly all SARS-CoV-2 samples worldwide.
D614G has been found to enhance viral replication in human lung epithelial cells and primary human airway tissues by increasing infectivity and stability of virons. Additional research has suggested that the increased infectivity may be the result of enhanced functional S protein assembly on the surface of the viron. Several other studies have reported that D614G may be associated with higher viral loads. Fortunately, since this mutation became common to nearly all sequenced SARS-CoV-2 genomes before the release of COVID-19 vaccines, we can be confident that vaccines with proved efficacy against SARS-CoV-2 are protective against the D614G mutation.
N-Terminus
The N-terminal S1 subunit of S protein is responsible for virus-receptor binding of SARS-CoV-2. Research indicates that acquisition of nucleotide deletions in the amino (N)-terminal domain (NTD) of the S protein may alter antigenicity. According to the Centers for Disease Control and Prevention (CDC), deletion of amino acids 69 and 70 in B.1.1.7, is likely to cause a conformational change in the spike protein. And creation of a Δ69Δ70 deletion mutant via site-directed mutagenesis and lentiviral pseudotyping resulted in 2-fold higher infectivity than the WT (D614G background), indicating that this linked pair of amino acid deletions may improve SARS-CoV-2 fitness.
Deletion of amino acid 144 in B.1.1.7 and amino acids 242-244 in B.1.351 have also been associated with reduced binding capacity of certain neutralizing antibodies.
Substitution of aspartate with glycine at position 253 (D253G), a mutation that appears in one of the 2 identified forms of B.1.526, has been correlated with escape from monoclonal antibodies against the NTD, as have L18F, a leucine (L) to phenylalanine (F) substitution at position 18 in P.1 and R246I, an arginine (R) to isoleucine (I) substitution at position 246 in B.1.351.
B.1.351, P.1, B.1.427/B.1.429. and B.1.526 all have additional amino acid substitutions in the NTD that are still of unknown significance.
Receptor Binding Domain (RBD)
The receptor binding domain (RBD) of S protein is comprised of amino acids 319-541. It binds directly to ACE2 receptors on human cells. Therefore, mutations in this portion of the genome are particularly significant to SARS-CoV-2 fitness and antigenicity.
B.1.1.7, B.1.351 and P.1 all have a mutation that replaces asparagine (N) with tyrosine (Y) at position 501 of the RBD. N501Y has been shown to increase binding capacity of SARS-CoV-2 to human ACE2 receptors, disrupt antibody binding to RBD and has been implicated in reduced antibody production via weakened T and B cell cooperation. Together, these findings suggest that SARS-CoV-2 variants possessing the N501Y mutation may have an increased potential for immune escape.
B.1.351 and P.1 have 2 additional RBD mutations in common, K417N or K417T, a lysine (K) to asparagine (N) or threonine (T) substitution at position 417, and E484K, a glutamate (E) to lysine (K) substitution at position 484. E484K increases the affinity of RBD for ACE2, increases resistance to SARS-CoV-2 neutralizing antibodies, is less responsive to monoclonal antibody therapy and reduces neutralization against convalescent plasma. Studies have demonstrated that, in combination, these 3 RBD mutations induce a relatively high conformational change, compared to N501Y alone or the WT strain, indicating increased potential for immune escape.
As mentioned above, B.1.526, has been detected by West Jr. et al. in 2 forms. One of these contains E484K, while the other contains S477N, a serine (S) to asparagine (N) substitution at position 477 that has also been shown to increase receptor binding affinity, suggesting that both forms may demonstrate increased viral infectivity.
The variant introduced by Zhang et al. as CAL.20C has also been detected in two forms, B.1.427 and B.1.429, both of which contain the same 3 S gene mutations that are not present in B.1.1.7, B.1.351, P.1 or B.1.526. One of these, L452R, is a substitution that replaces leucine (L) with arginine (R) at position 452 of the RBD and increases the affinity of RBD for ACE2. Reports of a study conducted by Chui et al. at USCF, indicate that B.1.429 is less susceptible to neutralizing antibodies and may be linked to worse outcomes of disease.
Furin Cleavage Site
The furin cleavage site of S protein subunits S1 and S2 is essential for membrane fusion of SARS-CoV-2. Loss of this structure or function has a major negative impact on the pathogenesis of the virus. B.1.1.7 has a proline (P) to histidine (H) substitution at position 681 that is located near the furin cleavage site. This mutation may further impact viral infectivity, although it is not yet clear if P681H enhances or decreases infectivity.
B.1.351 and B.1.526 both have an alanine (A) to valine (V) substitution located adjacent to the furin cleavage site at position 701 (A701V) that is still of unknown significance.
Hodcroft et al. have identified a rapid rise of SARS-CoV-2 infections that possess a substitution at position 677 of the S gene. Researchers suspect that proximity of the mutation to the furin cleavage site may impact the virus' ability to enter host cells, and parallel evolution in multiple lineages could suggest a selective advantage to the virus. So far, one sub-lineage carrying Q677P, a glutamine (Q) to proline (P) substitution, and at least 6 distinct sub-lineages carrying Q677H, a glutamine (Q) to histidine (H) substitution have been detected, demonstrating the importance of continued research to evaluate these S:677 polymorphisms.
C-Terminus
The C-terminal S2 subunit of S protein promotes virus-cell membrane fusion. B.1.1.7 and P.1 have multiple mutations in the C-terminal domain of S protein that are still of unknown significance.
Evaluating the Antigenicity of SARS-CoV-2 Variants
Amidst questions about antigenicity, researchers have begun to evaluate the neutralization potency of sera from vaccinated individuals against SARS-CoV-2 variants. Let’s first take a look at some data evaluating S gene mutations in the B.1.1.7 lineage, including the N501Y substitution.
B.1.1.7
On Jan. 8, 2021, Pfizer published a press release announcing that their vaccine elicited antibodies capable of neutralizing SARS-CoV-2 with the N501Y mutation in the lab. Results from this study, which was conducted by Pfizer in collaboration with the University of Texas Medical Branch (UTMB), were posted in a preprint one day before the press release was published. Researchers generated an isogenic Y501 mutant on the genetic background of the N501 clinical strain that was used to develop their vaccine. Then they tested the neutralization of both N501 and Y501 against sera from 20 vaccine trial participants, drawn 2-4 weeks after administration of the second dose of vaccine, and found no biologically significant difference in the neutralization activity against the Y501 mutant.
Another study tested the sera of 16 individuals who participated in Pfizer’s phase 1/2 trial in Germany against VSV-SARS-CoV-2-S pseudoviruses representing the WT Wuhan strain and the full B.1.1.7 lineage. Serum was drawn 21 days after administration of the second dose of vaccine. Once again, researchers found no biologically significant difference in the neutralization activity against the 2 pseudoviruses.
However, another preprint evaluated induced antibody responses of 23 participants, 3 weeks after the first dose of Pfizer vaccine, against lentiviral pseudotyped WT and B.1.1.7 virus lineages and found that neutralizing titers were reduced 3.85-fold against B.1.1.7 compared to WT. The addition of the E484K mutation, found in B.1.351 and P.1, induced further loss of neutralizing activity.
B.1.351
This preprint tested the neutralization potency of plasma taken from 20 volunteers 8 weeks after the second dose of either the Moderna or Pfizer vaccine against a panel of 10 mutant pseudotyped viruses. Researchers found that neutralizing activity was reduced by a small but significant margin against N501Y and the K417N:E484K:N501Y combination mutants.
Moderna also conducted its own study, in which researchers tested sera from Phase 1 trial participants against recombinant VSV-based SARS-CoV-2 pseudovirus neutralization assay from the WT (Wuhan) isolate, D614G variant, B1.1.7 and B.1.351 variants. They also tested the neutralization of sera from non-human primates and found no significant impact on neutralization against B.1.1.7 in either case. However, pseudoviruses with spike K417N-E484K-N501Y-D614 and the full B.1.351 mutations showed 2.7 and 6.4-fold reduction in neutralization, respectively. Importantly, even with this reduced activity, the serum that was tested was still able to fully neutralize the B.1.351 variant.
When Moderna and Pfizer announced the results of their phase 3 efficacy trials in Nov. 2020, public awareness about SARS-CoV-2 variants was just emerging. But Johnson & Johnson (J&J), who announced the interim analysis of its phase 3 efficacy trials in late Jan 2021, was able to design its trials to test vaccine efficacy against circulating SARS-CoV-2 variants like B.1.1.7 and B.1.351. The regional prevalence of particular variants may have contributed, at least in part, to the variability of data reported from the countries that participated in these trials (72% efficacy in the U.S., 66% in Latin America and 57% in South Africa). But in preparation for review of the company's Emergency Use Authorization (EUA) request in February, J&J presented new documents to the U.S. Food & Drug Administration (FDA) claiming the vaccine was 81.7% effective at reducing severe disease and 64% effective against moderate disease in South Africa, starting 28 days after vaccination.
Highlighting the Need for Genomic Surveillance
In summary, studies indicate that the B.1.1.7 variant is, in fact, neutralized by antibodies developed in individuals who have been vaccinated with Pfizer and Moderna mRNA vaccines. Reduced neutralization potency against the B.1.351 variant has been observed. However, studies have shown that serum of mRNA vaccinated individuals is still able to fully neutralize the B.1.351 variant. And J&J, the 3rd vaccine to receive EUA in the U.S., has still shown high efficacy against severe disease in individuals infected by B.1.1.7 and B.1.351. In order to ensure maximum efficacy of its product, Moderna announced that it has launched a clinical program to develop vaccine boosters that will account for current and emerging SARS-CoV-2 variants, and Pfizer has recently followed suite.
As always, it’s important to remember that this data is preliminary. Research evaluating neutralization potency against the P.1, B.1.427/B.1.429 and B.1.526 lineages is still needed, and new information about SARS-CoV-2 variants is being produced daily. Notably, B.1.1.7, B.1.351 and P.1 have now all been identified in multiple countries, including the United States, and because mutation is regularly occurring, new variants will continue to emerge. This ongoing mutation threat emphasizes the necessity of genomic surveillance programs that will track SARS-CoV-2 evolution, help contain the spread of disease and inform public health practices, including diagnostics and vaccine development and distribution.
AUTHOR : ASHLEY HAGEN, M.S.
Ashley Hagen, M.S. is the Science Communications Specialist at the American Society for Microbiology.
The original text is available on the website below.
Source : https://asm.org/Articles/2021/February/SARS-CoV-2-Variants-vs-Vaccines



